Home / News / India News / Article /
Dr. Reddy's Laboratories gets authorisation for Sputnik V in India
Updated On: 13 April, 2021 11:41 AM IST | New Delhi | IANS
This is as per the provisions of the New Drug and Clinical Trials rules, 2019 under the Drugs and Cosmetics Act.
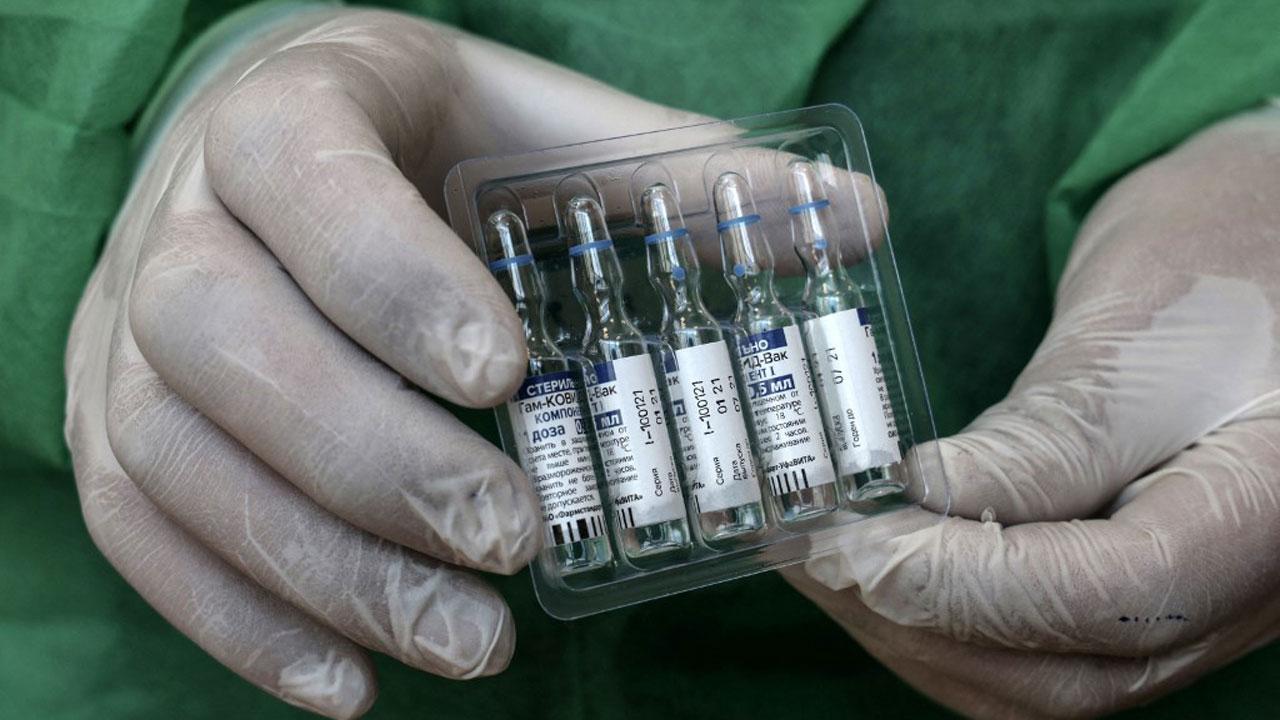
Vials of the Russian Sputnik V vaccine. Photo/AFP
Dr. Reddy's Laboratories Ltd. has announced that it has received the permission from the Drug Controller General of India (DCGI) to import the Sputnik vaccine into India for restricted use in emergency situations.
This is as per the provisions of the New Drug and Clinical Trials rules, 2019 under the Drugs and Cosmetics Act. In September 2020, Dr. Reddy's had partnered with the Russian Direct Investment Fund (RDIF) to conduct the clinical trials of Sputnik V and distribute the vaccine in India. In addition to the trials conducted in Russia by RDIF, Phase II / III clinical trials of the vaccine were carried out by Dr. Reddy's in India.



